Ever since life events made it impossible for me to maintain two blogs (this one and my not-so-super-secret other blog, which other than a handful of original posts has been mostly fallow for several months), one frustration that I’ve found with once-a-week blogging in the age of President Donald Trump and his Health and Human Services (HHS) Secretary Robert F. Kennedy, Jr.‘s “make America healthy again” (MAHA) is that there’s just too damned much material. Every week, since January I have a truly difficult time choosing between multiple horrors to write about. Sometimes the choice is obvious, such as when the CDC Advisory Committee on Immunization Practices (ACIP) meeting led to last week’s post about how RFK Jr. and the Trump administration will invoke a distorted version of “best practices” of other countries that don’t recommend vaccination against as many diseases as the US does to justify rolling back as many vaccine requirements as possible or my post from the week before writing about the director of the FDA Center for Biologics Evaluation and Research (CBER), Dr. Vinay Prasad, sending an email memo blaming ten pediatric deaths on COVID-19 vaccines without actually showing the work that led to that conclusion or citing the evidence, all while telling anyone who disagrees with his approach that they should resign. This time around, the choice was less obvious, with multiple horrors continuing to best science-based medicine and health policy, but one post on X, the hellsite formerly known as Twitter, led me to revisit a longtime topic that we at SBM haven’t written much about in a while. Does anyone remember the Dietary Supplement Health Education Act (DSHEA) of 1994?
MAHA does:
The DSHEA disclaimer, by the way, is something that, back in the day, we used to refer to as the “quack Miranda warning,” as I couldn’t resist taking to the same hellsite to mention:
I could have discussed more about the MAHA war on vaccines, but then I would neglect another RFK Jr. action that, in addition to being consistent with MAHA, is also consistent with the Trump administration’s deregulatory agenda and therefore much more likely to slide in under the radar, unlike RFK Jr.’s war on vaccines. I realize that some of this might come across as too much “inside baseball” to some of our readers, but it’s totally been in our wheelhouse here at SBM since the very beginning 18 years ago. Moreover, there are plenty of others writing about RFK Jr.’s latest attacks on vaccines, but I haven’t seen much written (yet) about this. (Indeed, a search of X only finds two posts about it.)
To return to the topic at hand, the quack Miranda warning is our term for the DSHEA disclaimer, which generally reads, “This/these statement/statements has/have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.” Often there will be other optional flourishes, such as saying that these statements are for “informational purposes only” and/or “not intended as medical advice,” all with a “see your doctor” finale. If there’s one thing we’ve discussed here again and again over the years, it’s how the DSHEA weakened supplement regulation and how a quack Miranda warning on a website is a major red flag that it is likely selling quackery.
I always knew that, sooner or later, RFK Jr. and his MAHA minions whom he’s placed in key positions in the regulatory agencies of HHS would get around to dietary supplements, because of course they would. After all, just look at key MAHA allies. One great example is Dr. Peter McCullough, cardiologist turned COVID grifter during the pandemic and now the “chief scientific officer” for The Wellness Company, which, in addition to selling ivermectin to treat and prevent COVID-19, also sells various supplements to treat COVID-19 “vaccine injury.” Indeed, the wellness influencers who got their start during the pandemic have now become indistinguishable from the quacks of old who used to sell all manner of unproven (and disproven) supplements.

So what is the FDA doing about the quack Miranda warning? Before I discuss how the above proposal will weaken the already weak DSHEA labeling requirement, most likely as a first step towards weakening dietary supplement regulation further, let’s discuss what the DSHEA is (and is not) and how it set the stage for the proliferation of unproven supplements sold as medicine with a not-so-subtle, “wink-wink, nudge-nudge.” (It also provide a mechanism for huge revenue streams for quacks like Joe Mercola and right wing conspiracy theorist influencers, like Alex Jones.)
The DSHEA and the Quack Miranda warning
I’ve known that the quack Miranda warning would be on RFK Jr.’s target list ever since late summer of 2024, when, after having abandoned his independent campaign for President and bent the knee to Donald Trump in return for the position of HHS Secretary if Trump won, he published what I called a “MAHA manifesto” in the Wall Street Journal and then, a week before the election, declared MAHA war on the FDA thusly:

Of course, peptides and “nutraceuticals” are often sold as dietary supplements, complete with marketing claiming health benefits and the treatment of some diseases, as are some chelating compounds. It was at this point that it became clear to me that RFK Jr. had pivoted from being primarily antivaccine to embracing a more general “health freedom” orientation frequently associated with the antivaccine movement that paints the Food and Drug Administration as the pharma-serving villain suppressing “natural” treatments, low cost medications, and “miracle cures.” I also realized that I shouldn’t have been surprised by this open attack on the FDA, given that it’s long been a goal of the “health freedom” movement to dismantle the FDA and return it at least to a pre-thalidomide state if not to eliminate the FDA entirely. Recall that, before the thalidomide scandal, drug manufacturers were only required to submit evidence of safety, not efficacy, to win approval to market their products. After the thalidomide scandal, Congress passed the Kefauver-Harris Amendments in 1962. These Amendments to the federal Food, Drug, and Cosmetic Act tasked the FDA with additional requirements and responsibilities:
- Efficacy requirement: Manufacturers must provide substantial evidence that a drug is effective for its intended use before it can be sold.
- Retroactive review: The FDA was required to review the effectiveness of drugs approved between 1938 and 1962, a review that by the early 1970s had categorized approximately 600 medicines as “ineffective” and forced their removal from the market.
- Informed consent: The amendment mandated that subjects provide informed consent before participating in clinical trials.
- Good manufacturing practices: The amendments established rules for drug manufacturing and required routine inspections of production facilities.
- Adverse event reporting: Manufacturers were for the first time required to report serious side effects to the FDA.
- Advertising regulation: The amendments transferred the regulation of prescription drug advertising from the Federal Trade Commission (FTC) to the FDA and required more truthful and informative advertisements. Drug manufacturers were required to inform consumers of side effects and contraindications in their advertising.
You can see why the “health freedom” movement that ultimately spawned MAHA over six decades later did not like these amendments. But where does the DSHEA fit in? Basically, it’s a law that, by intent, made it difficult for the FDA, even when it wanted to, to act on dubious health claims for unproven, disproven, and sometimes even outright harmful supplements. As one of our former bloggers here at SBM, Dr. Peter Lipson, once described it long ago, the DSHEA is a “travesty of a mockery of a sham.”
According to the FDA‘s discussion of the DSHEA of 1994:
A dietary supplement is a product intended for ingestion that, among other requirements, contains a “dietary ingredient” intended to supplement the diet. The term “dietary ingredient” includes vitamins and minerals; herbs and other botanicals; amino acids; “dietary substances” that are part of the food supply, such as enzymes and live microbials (commonly referred to as “probiotics”); and concentrates, metabolites, constituents, extracts, or combinations of any dietary ingredient from the preceding categories.
There are two types of ingredients that may be used in dietary supplements: “dietary ingredients,” discussed above, and “other ingredients.” These “other ingredients” include substances such as fillers, binders, excipients, preservatives, sweeteners, and flavorings.
As you can see, one thing that the DSHEA does is to make a distinction between food and medicine. It also requires that dietary supplements be ingested and not injected or breathed in, which also makes sense. While on its surface distinguishing between food and medicine would seem to be quite reasonable (after all, it wouldn’t make a lot of sense to hold food to the same standards to which drugs are held), as implemented by the DSHEA this distinction has a pernicious effect in that in practice it allows manufacturers to label all sorts of botanicals, many of which with pharmacological activity, as “supplements,” and supplements, being defined as food and not medicine, do not require prior approval by the FDA before marketing:
FDA regulates both finished dietary supplement products and dietary ingredients. FDA regulates dietary supplements under a different set of regulations than those covering “conventional” foods and drug products. Under the Dietary Supplement Health and Education Act of 1994 (DSHEA):
- Manufacturers and distributors of dietary supplements and dietary ingredients are prohibited from marketing products that are adulterated or misbranded. That means that these firms are responsible for evaluating the safety and labeling of their products before marketing to ensure that they meet all the requirements of the Federal Food, Drug, and Cosmetic Act as amended by DSHEA and FDA regulations.
- FDA has the authority to take action against any adulterated or misbranded dietary supplement product after it reaches the market.
In other words, when a supplement is marketed it’s more or less the honor system. No registration with the FDA is required. After all, supplements are food, not medicine! (Just like “fish are friends, not food”?) Even though there are exceptions that require manufacturers to notify the FDA if they add a new ingredient to a supplement, in effect, the government can’t really do much of anything unless problems are reported after the supplement is marketed. Even worse, the definition of “supplement” has become very broad, as Quackwatch pointed out:
The Food, Drug, and Cosmetic Act defines “drug” as any article (except devices) “intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease” and “articles (other than food) intended to affect the structure or function of the body.” These words permit the FDA to stop the marketing of products with unsubstantiated “drug” claims on their labels.
To evade the law’s intent, the supplement industry is organized to ensure that the public learns of “medicinal” uses that are not stated on product labels. This is done mainly by promoting the ingredients of the products through books, magazines, newsletters, booklets, lectures, radio and television broadcasts, oral claims made by retailers, and the Internet.
DSHEA worsened this situation by increasing the amount of misinformation that can be directly transmitted to prospective customers. It also expanded the types of products that could be marketed as “supplements.” The most logical definition of “dietary supplement” would be something that supplies one or more essential nutrients missing from the diet. DSHEA went far beyond this to include vitamins; minerals; herbs or other botanicals; amino acids; other dietary substances to supplement the diet by increasing dietary intake; and any concentrate, metabolite, constituent, extract, or combination of any such ingredients. Although many such products (particularly herbs) are marketed for their alleged preventive or therapeutic effects, the 1994 law has made it difficult or impossible for the FDA to regulate them as drugs. Since its passage, even hormones, such as DHEA and melatonin, are being hawked as supplements.
In other words, under the DSHEA, substances that are clearly not foods can be marketed as supplements, including herbs and other botanicals (the vast majority of which are marketed as having a beneficial medicinal effect and some of which contain chemicals that do act as drugs).As long as the manufacturer is careful not to make specific health claims, it’s all good. In other words, a “nutritional support” statement claimed for a supplement must not be a “drug” claim; i.e., it must not claim that the supplement can be used for the treatment or prevention of a disease. Supplement manufacturers easily evade this requirement by making vaguer claims related to organs or systems, such as claiming that a product “boosts the immune system,” “supports heart health,” or something similar, often accompanied by what Dr. Lipson has sarcastically referred to as the “quack Miranda warning.“
Arguably, the DSHEA was the greatest boon to supplement manufacturers ever. In its wake, the supplement industry exploded, racking up huge increases in sales. Indeed, by 2007 a GAO report estimated that the supplement industry had grown to a $23.7 billion industry, and, in 2025, Grand View Research estimated that the size of the dietary supplement industry has reached $112.6 billion and also forecast it to grow at a compound annual growth rate (CAGR) of 4.9% from 2025 to 2030. Moreover, so lax was the regulation of supplements even before RFK Jr, and MAHA took over HHS, that it took a very extreme and egregious act, namely the marketing of an industrial chelator as an “antioxidant” supplement for the treatment of autism, before the FDA finally acted, and the FDA has seldom acted forcefully even over the one aspect of supplement regulation that the DSHEA does give it, to recall products due to “adulteration.”
Over the years, supporters of science-based medicine and sound public policy have made efforts in Congress to alter or repeal the DSHEA, such as the Dietary Supplement Safety Act of 2010, which would, among other things, have mandated reporting of all adverse events due to supplements to the FDA and given the FDA mandatory recall power over supplements that cause harm. All have failed to pass, as the DSHEA has had powerful patrons, such as its co-sponsors Sen. Tom Harkin (D-Iowa) and Sen. Orrin Hatch (R-Utah), and Utah is home to many supplement manufacturers. Sens. Harkin and Hatch might have retired from the Senate years ago, but they have many successors in Congress. With MAHA ascendant, supplement manufacturers in their stans are more powerful than ever, as is the Congressional Dietary Supplement Caucus. Yes, that’s a real thing, and, although Republicans do outnumber Democrats in the caucus, the caucus is currently pretty bipartisan, with a ratio of Republicans to Democrats of roughly 3:2.
Next, let’s look at what the FDA plans to do.
Too many quack Miranda warnings?
Let’s take a look at the text of the FDA letter to supplement manufacturers from Kyle Diamantas, Deputy Commissioner for Human Foods. The letter is dated December 11, and entitled Letter to the Dietary Supplement Industry on the DSHEA Disclaimer. The first thing I noted is that the letter starts out by stating that the FDA has “received several requests to amend our labeling regulation at 21 CFR 101.93(d),1 which governs the placement of the disclaimer required under section 403(r)(6)(C) of the Federal Food, Drug, and Cosmetic Act (FD&C Act).” Specifically, the FDA has required that the DSHEA disclaimer (a.k.a. the quack Miranda warning) be placed on every panel in supplement documentation in which a health claim is made.
According to the letter, currently the requirement mandates:
This regulation states the DSHEA disclaimer shall be placed adjacent to the statement with no intervening material or linked to the statement with a symbol (e.g., an asterisk) at the end of each statement described in section 403(r)(6) of the FD&C Act, such as a structure/function claim, (i.e., a “403(r)(6) claim”). The regulation goes on to state “the disclaimer shall appear on each panel” of a product label where there is a 403(r)(6) claim.
A “structure-function” claim is another way that the DSHEA allows supplement manufacturers to bypass the requirement that they cannot claim to treat or prevent any diseases. You’ve seen these sorts of structure-function claims before. Examples include:
- “Supports [specific organ] health.” Examples include: “supports prostate health”; “supports brain health”; “supports immune health”; “helps maintain a healthy heart.”
- The ever-popular “boosts the immune system” or “supports immunity”
- “Builds strong bones”
A study from several years ago found that 87% of advertisements for supplements contain structure-function claims, with an average of 2.5 claims per ad. There is a great PowerPoint presentation from Venable, LLP outlining the differences between permissible and impermissible structure-function claims based on eleven (!) FDA criteria. If you peruse the list, you’ll find that some of these are truly splitting hairs, like, for example, these two (click to embiggen):



In any event, regardless of the structure-function claim, there has to be at least an asterisk leading to a quack Miranda warning for every instance, at least under current FDA regulations.
So what does the FDA now want to do? Back to the letter:
Based on FDA’s initial review, we expect that revising this regulation to remove the requirement for the DSHEA disclaimer to appear on each panel of a product label where there is a 403(r)(6) claim would be consistent with section 403(r)(6)(C) of the FD&C Act while reducing label clutter and unnecessary costs. Further, FDA recognizes that we have rarely, if ever, enforced this requirement. If FDA does not identify significant concerns as we continue our review of the available data and information regarding this request, we are likely to propose a rule to amend this requirement.
I like how Diamantas basically admits that the FDA wasn’t really enforcing this requirement very much anyway and the goes on to say that the FDA will exercise “enforcement discretion regarding the requirement for the DSHEA disclaimer to appear on each panel of a product label where a 403(r)(6) claim appears,” while still requiring that the DSHEA disclaimer appear at least once.
Why, you might ask, would the supplement manufacturers want this regulation changed so badly? One obvious answer, of course, is that if the quack Miranda appears only once in the product’s labeling it will be very easy for consumers to overlook it, as opposed to when it is required to appear on every panel in which a structure-function claim is made. There’s another reason, though, that was revealed in the congratulatory messages posted by supplement manufacturing groups, such as the Council for Responsible Nutrition (CRN), which “applauded FDA clarification on DSHEA disclaimer after years of industry advocacy“:
For nearly 30 years, FDA practice has allowed the use of an asterisk to direct consumers to the full DSHEA disclaimer elsewhere on the label, consistent with section 403(r)(6)(C) of the Food, Drug & Cosmetic Act and 21 C.F.R. § 101.93(d). However, a recent uptick in class-action lawsuits has relied on an alternative interpretation, arguing that the full disclaimer must be placed on every panel where a structure/function claim is used on a product label. These legal challenges have created confusion, prompted unnecessary litigation, and imposed costly and disproportionate burdens on responsible manufacturers—without improving consumer understanding.
Earlier this year, CRN called on FDA [starting on page 23] to revise section 101.93(d) to clarify that the law does not require duplicative use of the DSHEA disclaimer on multiple panels of the same product. CRN offered then that making the change “would maintain adequate levels of consumer protection while reducing unnecessary, redundant regulatory burdens on companies,” and argued that a single disclaimer on a dietary supplement product’s label linked by an asterisk or other symbol to structure/function claims adequately informs consumers of the existence of additional information and satisfies the legislative intent to have a structure/function statement “contain” the disclaimer. Separately, CRN made clear to the agency that it was prepared to file a citizen petition to formally escalate the matter if needed.
Those pesky citizen activists and their class-action suits, getting in the way of supplement industry obfuscation about its products! Make no mistake, the supplement industry sees this as a big win and a harbinger of things to come. Here’s the American Herbal Products Association’s take:
This significant action comes directly in response to sustained advocacy from AHPA dating back to and continuing since 2021. AHPA and others have argued that the existing “every panel” requirement (found in 21 C.F.R. § 101.93(d)) is overly prescriptive and exceeds the demands of DSHEA. AHPA has long advocated for FDA to update the regulation to reflect historical industry practice: prominently displaying the disclaimer once on the product label or labeling, not necessarily on every panel, and connecting each claim to the disclaimer via symbols (e.g., asterisks). The contemplated revision would also accord the technical requirements of the regulation with FDA’s historical enforcement approach; as conceded in the letter, and consistent with AHPA’s analysis of agency documents, FDA has “rarely, if ever, enforced this requirement.”
“This news is a major win for the dietary supplement industry,” said AHPA President & CEO Graham Rigby. “AHPA has long led the charge for this sensible regulatory reform, and we thank FDA for taking action on an unenforced provision that has fueled opportunistic litigation. This is a strong, positive signal of the agency’s commitment to modernizing regulations, and AHPA looks forward to continued engagement in shaping a rational and modern regulatory environment for this vital category of products.”
Again, I realize that this might strike some readers as too much “inside baseball,” but there are real consequences. Some readers might reasonably wonder why it matters if the quack Miranda warning appears on every panel where disease treatment claims barely disguised as “structure-function” claims or not. I found the most succinct explanation here:
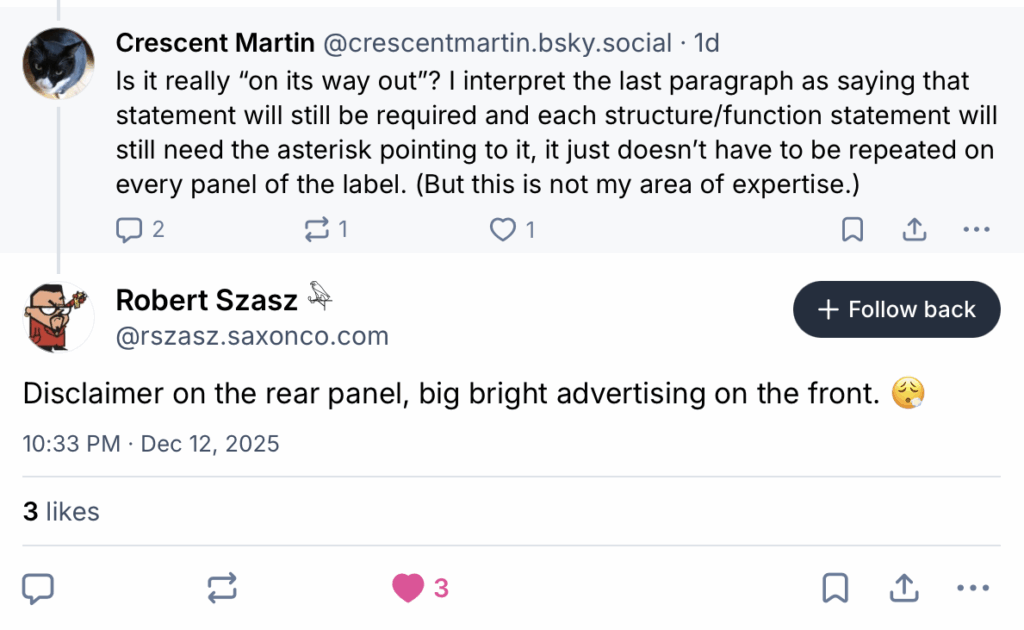
Moreover:
The current relevant regulations state:
The disclaimer shall be placed adjacent to the statement with no intervening material or linked to the statement with a symbol (e.g., an asterisk) at the end of each such statement that refers to the same symbol placed adjacent to the disclaimer specified in paragraphs (c)(1) or (c)(2) of this section. On product labels and in labeling (e.g., pamphlets, catalogs), the disclaimer shall appear on each panel or page where there such is a statement. The disclaimer shall be set off in a box where it is not adjacent to the statement in question.
Getting rid of the requirement for the DSHEA on each panel will make it a lot easier for supplement manufacturers to hide the “prominent” disclaimer, to bury it somewhere else and then refer to it by an asterisk.
What does this mean, and does it matter?
As I mentioned before, I realize that to many people this change seems minor. Indeed, one can argue if DSHEA disclaimers (or, as we prefer to call them, quack Miranda warnings) even matter that much to consumers, if they dissuade them from purchasing a product. After all, Cheerios claims to help maintain a healthy cholesterol in its ads, along with a quack Miranda warning. This is about more than just supplement manufacturers. Many food manufacturers use the DSHEA as well to justify structure-function health claims for their products.
Nonetheless, given RFK Jr.’s declaration of MAHA war on the FDA, I nonetheless believe that this proposed change is important for a number of reasons. First, it signals to the supplement industry that RFK Jr. is on its side now and that the FDA will be far more compliant in terms of doing the industry’s bidding than ever before. Second, this step is clearly meant as a first step in deregulating supplements using the most industry-friendly interpretation of the DSHEA that RFK Jr. and his cronies can come up with. The next steps, I predict, will be to loosen the restrictions on what is considered a “structure-function” claim, so that supplement manufacturers can make more explicit disease treatment and prevention claims without the evidence that would be required for a manufacturer of a drug to make a similar claim.
Finally, as Tim Caulfield, whose X post was quoted above notes, one only has to look at the regulatory double standard in terms of evidence required between supplements, for which RFK Jr. clearly plans to lower the bar as much as possible, and vaccines to see what MAHA is really about. News reports now indicate that, based on a non-transparent “analysis” of pediatric deaths linked with COVID vaccines that has only been referred to thus far in a leaked memo and blames the vaccines for killing at least ten children without explaining how the FDA came to the conclusion of causation, the FDA plans on adding a black box warning to COVID vaccines. A “black box” warning is the FDA’s strongest warning included in the package insert for a drug, medical device, vaccine or biological. Usually such warnings are about a significant risk of death or serious injury due to a product that must be weighed against the product’s benefit. Sometimes such warnings are targeted to certain groups most at risk, for example the warning for Accutane of potential birth defects when used during pregnancy. It’s also interesting to note how the FDA is going through the standard regulatory process to change the requirements for how a quack Miranda warning must be displayed but is, according to news reports, not doing so with its changes to the package insert for COVID vaccines:
“I guess my concern is that in this case, there’s not a process,” said Kesselheim, who has studied the impact of black box warnings on drug labels. “There isn’t that same opportunity for discussion and good-faith review of the data on which this decision is being made.
“My concern is that this will be seen as yet another in the long line of decisions that this FDA is making based on its own political goals, rather than after a dutiful and thoughtful and public review of the science.”
When I first noted that RFK Jr. is coming for your vaccines, I outlined six strategies that he would use. Strategy #6 read: Co-opt the FDA to make approval of new vaccines more difficult and issue black-box warnings based on antivax fear mongering for existing vaccines. (I hate to say I told you so, but…) I further noted that RFK Jr. would, through his minions, cherry pick and misrepresent evidence to portray vaccines as dangerous in order to accomplish this.
This double-standard is inherent in MAHA: Massively lower standards for unproven treatments that “health freedom” and wellness influencers like and profit from, such as supplements, quack treatments, unproven stem cell treatments, and the like, while increasing the regulatory bar for vaccines and certain pharmaceuticals to impossible levels, with the goal being to ultimately remove them from the market. Adding a black box warning to COVID vaccines is just one step.
I hate to end without a bit of hope. As we all know, the Trump administration is all about deconstructing the administrative state and deregulating favored industries, using Project 2025 as a playbook, the better to turn staffing the government back to the spoils system, one manifestation of which is how MAHA wants to turn the criteria for NIH funding of research away from pure scientific merit and towards patronage and political loyalty, the better to make it a tool to enforce ideological conformity. There is one area in which this deregulatory drive is running afoul of MAHA activists, specifically the dismantling of the Environmental Protection Agency, which is angering “MAHA moms,” as reported in the New York Times over the weekend, to the point that EPA Administrator Lee Zeldin recently met with a group of very unhappy MAHA moms who are accusing the agency of prioritizing chemical industry interests over public health through its efforts to loosen restrictions on harmful chemicals, and approve new pesticides, loosen pollution regulations, not to mention Trump’s having appointed several former chemical industry executives and lobbyists to senior roles overseeing pesticides and toxic chemicals.
Of course, as I pointed out last week, this was always part of the Faustian bargain RFK Jr. made when he bent the knee to Donald Trump: Abandon the environmentalist activism for which he was primarily known before turning antivax activist in return for a chance at getting rid of as many vaccines as he can. When it comes to loosening restrictions on supplements while ratcheting up restrictions on vaccines to untenable levels, there is no conflict. The same might not be true when it comes to being complicit in the extreme deregulatory agenda being promoted at the EPA. I suspect that not even RFK Jr. can square that circle indefinitely. At least I hope not.
Unfortunately, in the meantime, massive damage has already been done to the vaccine program and the regulatory apparatus that assures safe and effective drugs, with over three years for this administration to do worse.

